Rare Disease Clinical Trials Market is expected to reach US$ 30.13 Billion by 2033 | DataM Intelligence
The Global Rare Disease Clinical Trials Market is expected to reach at a CAGR of 9.39% during the forecast period 2026 to 2033.
The Global Rare Disease Clinical Trials Market is rapidly expanding, driven by growing rare disease research, regulatory support and innovation, targeting significant unmet needs.”
AUSTIN, TX, UNITED STATES, March 10, 2026 /EINPresswire.com/ -- Market Overview: — DataM Intelligence
The Global Rare Disease Clinical Trials Market has witnessed significant growth over the past decade, driven by the increasing prevalence of rare diseases and advancements in biotechnology and personalized medicine. Rare diseases, often defined as conditions affecting a small fraction of the population, pose unique challenges in diagnosis, treatment, and drug development. As a result, pharmaceutical companies and research organizations are investing heavily in clinical trials that target these niche patient populations. These trials are crucial not only for developing orphan drugs but also for ensuring regulatory compliance and accelerating market approvals in key regions.
To Download Sample Report Here: https://www.datamintelligence.com/download-sample/rare-disease-clinical-trials-market
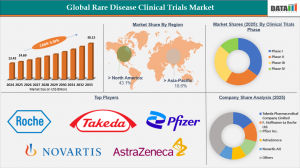
According to DataM Intelligence, The Rare Disease Clinical Trials Market is projected to reach USD 30.13 billion by 2033, growing at a compound annual growth rate (CAGR) of 9.39% from 2026 to 2033. The primary growth drivers include increasing government support for orphan drug development, rising awareness about rare diseases, and advances in gene therapies and molecular diagnostics. Among the various segments, biologics-based trials hold a leading position due to their potential to provide targeted therapies for genetic and metabolic disorders. Geographically, North America dominates the market, owing to well-established clinical research infrastructure, favorable regulatory frameworks, and substantial funding from both private and public sectors.
Key Highlights from the Report:
North America leads the market in terms of clinical trial investments and patient recruitment.
Biologics and gene therapy-based trials account for the largest market share in product type.
The market is projected to grow at a CAGR of 9.39% between 2026 and 2033.
Increasing patient advocacy programs are accelerating clinical trial awareness and participation.
Emerging economies in Asia-Pacific are witnessing rapid adoption of rare disease trials due to improved healthcare infrastructure.
Regulatory incentives, including orphan drug designation and tax credits, are driving innovation in the market.
Market Segmentation:
The Rare Disease Clinical Trials Market can be segmented based on product type, therapeutic area, and end-user. By product type, the market primarily includes biologics, small molecules, gene therapies, and combination therapies. Biologics remain the leading segment due to their ability to target rare genetic disorders effectively, while gene therapies are gaining traction because of their potential for curative outcomes.
In terms of therapeutic areas, oncology-related rare disease trials dominate, followed by metabolic and neurological disorders. These areas are prioritized due to the high unmet medical need and potential for breakthrough treatments. End-users in the market comprise pharmaceutical and biotechnology companies, contract research organizations (CROs), academic and research institutions, and government agencies. Pharmaceutical and biotechnology firms lead the market as they drive most clinical trial initiatives, supported by CROs that provide essential trial management services.
Speak to Our Analyst and Get Customization in the report as per your requirements: https://www.datamintelligence.com/download-sample/updated/rare-disease-clinical-trials-market?sz
Regional Insights:
North America remains the largest market for rare disease clinical trials, fueled by extensive research funding, a strong presence of key pharmaceutical companies, and well-established regulatory pathways. The United States, in particular, benefits from the Orphan Drug Act, which provides incentives such as market exclusivity, tax credits, and grant programs.
Europe is also a significant market, with countries like Germany, France, and the UK actively promoting rare disease research through initiatives such as the European Union’s Horizon programs. The region’s collaborative networks, patient registries, and robust healthcare systems make it conducive for multi-center trials.
Asia-Pacific is emerging as a high-growth region due to increasing investment in healthcare infrastructure, growing patient populations, and favorable regulatory reforms in countries such as Japan, China, and India. Clinical trial outsourcing to this region is becoming more common because of lower operational costs and a rapidly expanding skilled workforce.
Market Dynamics:
Market Drivers
The growth of the Rare Disease Clinical Trials Market is primarily driven by rising prevalence of rare diseases globally and increasing investments in orphan drug development. Government initiatives, such as financial incentives, regulatory support, and public-private partnerships, are accelerating clinical trial activities. Additionally, advances in gene editing, molecular diagnostics, and personalized medicine have enabled the development of innovative therapies, creating new opportunities for market expansion.
Market Restraints
Despite the growth potential, the market faces challenges, including high research and development costs, limited patient populations, and complex regulatory requirements across regions. Recruitment and retention of participants in rare disease trials remain a critical bottleneck, often extending trial timelines and increasing overall expenditure.
Market Opportunities
Emerging technologies, such as AI-driven patient recruitment, decentralized trials, and real-world data analytics, present significant opportunities to optimize trial design and execution. Additionally, increasing collaborations between pharmaceutical companies, CROs, and patient advocacy groups can enhance trial efficiency and reduce operational risks, further supporting market growth.
Looking For Full Report? Get it Here: https://www.datamintelligence.com/buy-now-page?report=rare-disease-clinical-trials-market
Frequently Asked Questions (FAQs):
How big is the Rare Disease Clinical Trials Market globally?
Who are the key players in the Rare Disease Clinical Trials Market?
What is the projected growth rate of rare disease clinical trials?
What is the market forecast for rare disease clinical trials in 2032?
Which region is estimated to dominate the Rare Disease Clinical Trials Market through the forecast period?
Company Insights:
Key players in the Rare Disease Clinical Trials Market include:
Takeda Pharmaceutical Company Limited
F. Hoffmann-La Roche Ltd.
Pfizer Inc.
AstraZeneca
Novartis AG
Labcorp
IQVIA
ICON plc
Parexel International (MA) Corporation
Syneos Health
Recent Developments:
United States:
February 2026: FDA launched a framework to accelerate individualized therapies for ultra-rare diseases, emphasizing disease-causing variant identification.
February 2026: FDA introduced a "plausible mechanism pathway" to broaden gene-editing successes for rare disease treatments.
January 2026: FDA dropped certain manufacturing quality control requirements for cell and gene therapies targeting unmet needs in rare diseases, easing clinical development paths.
January 2026: FDA implemented adaptive clinical trial allowances alongside prior Rare Disease Evidence Principles from September 2025, enabling approvals from single-arm studies.
Japan:
March 2026: Japan's MHLW granted orphan drug designation to rilzabrutinib specifically for IgG4-related disease, recognizing unmet needs.
February 2026: Sanofi's rilzabrutinib (Wayrilz) received orphan drug designation from MHLW for IgG4-related disease, supporting rare disease therapy advancement.
December 2025: DDrare updated medical expenses recipients data to FY2024, enhancing clinical trial insights for intractable diseases.
December 2025: DDrare database featured publication in Jikken Igaku and poster presentation at the Molecular Biology Society of Japan meeting on rare disease drug development.
Unlock 360° Market Intelligence with DataM Subscription Services: https://www.datamintelligence.com/reports-subscription
Conclusion:
The Rare Disease Clinical Trials Market is poised for robust growth, fueled by rising prevalence of rare diseases, technological advancements, and favorable regulatory support. Biologics and gene therapies continue to dominate as preferred treatment modalities, while North America remains the leading regional market. With emerging opportunities in Asia-Pacific and increasing collaboration among key stakeholders, the market is expected to evolve rapidly, offering innovative solutions for patients with unmet medical needs. Stakeholders, including pharmaceutical companies, CROs, and research institutions, are strategically positioned to capitalize on this growth trajectory, shaping the future of rare disease therapeutics.
Related Reports:
Rare Disease Therapeutics Market
Oncology Clinical Trials Market
Sai Kiran
DataM Intelligence 4Market Research
877-441-4866
Sai.k@datamintelligence.com
Visit us on social media:
LinkedIn
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.